Our Products
Our product portfolio supports and manages the packaging and print processes for brand owners, retailers, designers, packaging manufacturers and print service providers.

Content & Collaboration
Discover seamless content creation and collaboration by simplifying workflows, enhancing creativity, and achieving unparalleled efficiency.
Structure & Graphics
Enhance your visual communication, packaging, and design processes for exceptional quality and impactful results.
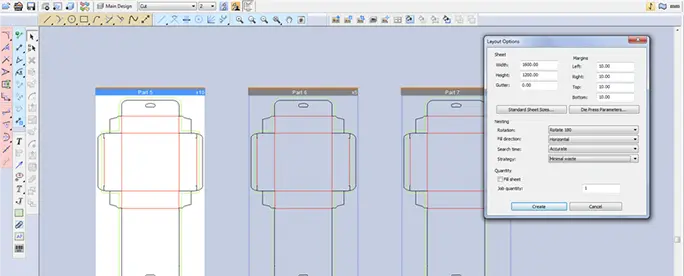
ArtiosCAD
Structural Packaging Design
Create 2D and 3D structural designs for product development, virtual prototyping, and more.

Studio
3D Packaging Design Software
Produce better packaging artwork in Adobe® Illustrator®.
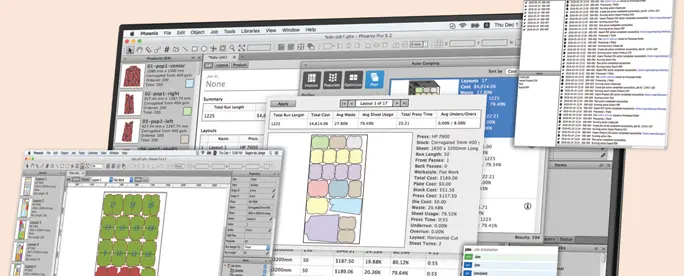
Phoenix
AI-Powered Planning & Imposition
Automated preproduction software for packaging manufacturers.

ArtPro+
PDF Prepress Editing
A native PDF editor for packaging prepress that easily prepares artwork for printing production.
DeskPack
Prepress Plugins for Adobe® Creative Cloud applications
Transform Adobe® Creative Cloud applications into full-fledged packaging prepress tools.
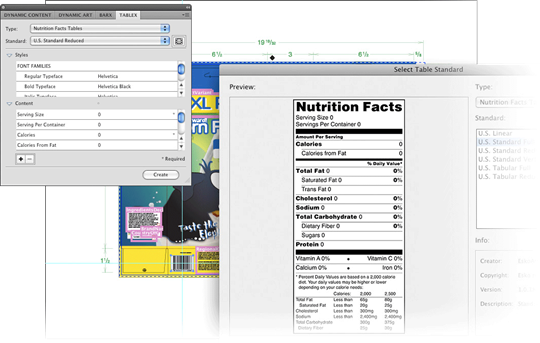
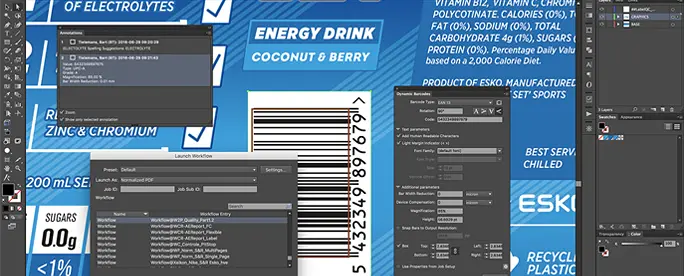
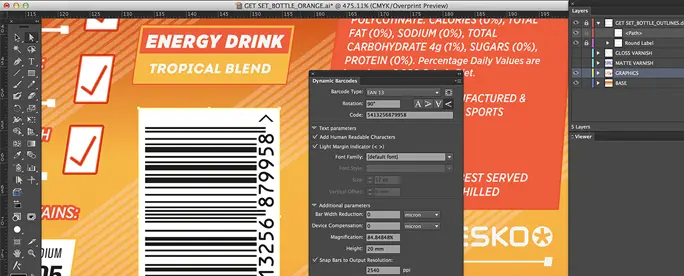
Dynamic Content
Text Management Plugin for Adobe® Illustrator®
Use Dynamic Content to avoid errors by separating packaging content from design.
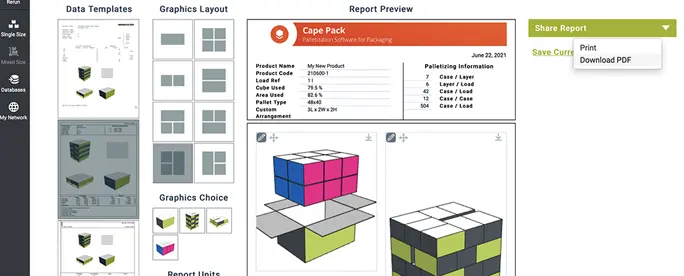
Cape Pack
Palletization Software
Determine the best product size, case count, product arrangement, case size, pallet, and container load for shipping – all while reducing environmental impact. Pack smarter. Ship greener.

Cape Truckfill
Container Loading Software
Optimize your packaging supply chain and reduce costs.

Store Visualizer
Virtual Reality Software
Test the visual impact of your latest design in a virtual store.
Color & Print
From design to print, optimize your color management and printing processes for accurate color reproduction, improved quality, and stunning visuals.
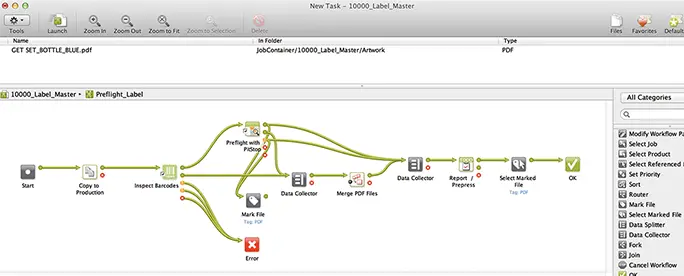
Automation Engine
Prepress Workflow Software
Automate workflows across various printing techniques such as digital, flexo, offset, and more.
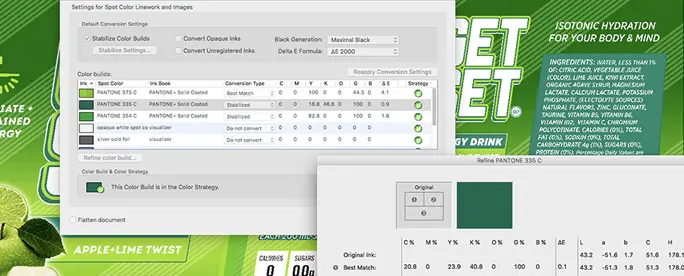
Equinox
Extended Gamut Printing
Achieve more and better colors by adding extra colors to the traditional CMYK range.

Imaging Engine Essentials
Raster Image Processor (RIP)
Tailored screening and calibration tools for optimal CDI performance.
Flexo Platemaking

CDI Quartz
Market-Leading Flexo Print Quality
Produce the most accurate, highest-quality plates and reduce ink usage, informed by AI-driven imaging insights that strengthen overall print quality.

CDI Crystal
Flexo Plate Imaging
Create flawless plates that excel in high print output and press stability.

CDI Spark
Flexo Plate Imagers
Caters to small & large plates, tag & label, flexible packaging, folding carton, & corrugated segments.

XPS Crystal
Flexo Plate Exposure Unit
Unlock the ability to produce highly consistent digital flexo plates, every time.

CDI Crystal XPS
Combined Flexo Plate Imager and Exposure Unit
Get the perfect, consolidated flexo platemaking workflow.

HD Letterpress
Imaging for Metal-Backed Plates
Tackle dry offset printing challenges and ensure smooth highlights and precise linework.
Get Started with Esko
Contact us today to learn more about our products and services.