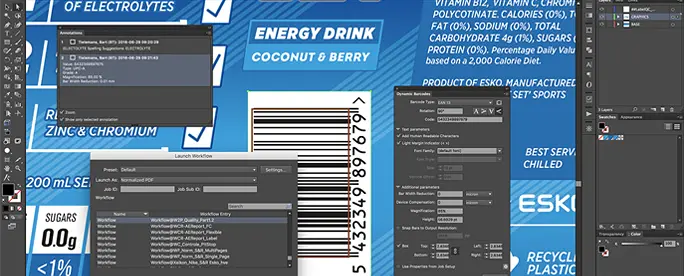
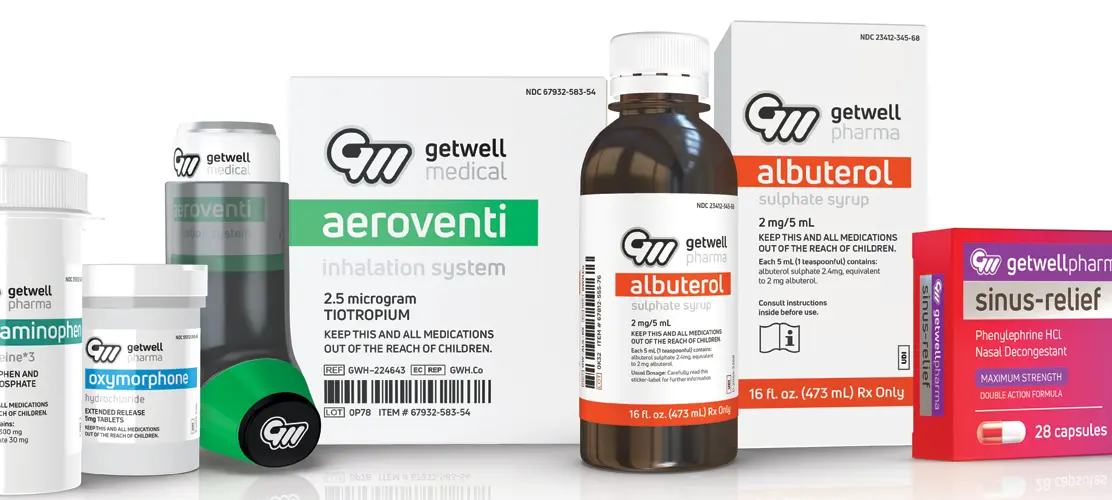
Built to Meet Life Sciences Packaging Demands
The Essential Guide to Pharma Labeling Excellence Using Structured Content Management
Learn how labeling, artwork management, and packaging can help your company enhance efficiency and maximize results.
Featured Products for Life Sciences
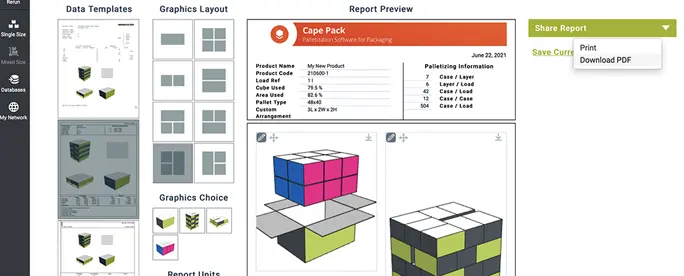
Cape Pack
Palletization Software
Determine the best product size, case count, product arrangement, case size, pallet, and container load for shipping – all while reducing environmental impact. Pack smarter. Ship greener.
ArtiosCAD
Structural Packaging Design
Create 2D and 3D structural designs for product development, virtual prototyping, and more.

Studio
3D Packaging Design Software
Produce better packaging artwork in Adobe® Illustrator®.