Written by Gouri Sasidharan
Content Specialist, Esko
Among product recalls, shockingly, nearly 40% stem from labeling errors that impact compliance with overall packaging requirements.
Here’s the uncomfortable truth: these issues are preventable, but only when the compliance process itself is structurally sound.
If your labeling audit preparation checklist still lives in spreadsheets, regulatory approvals move through long email threads, or artwork files sit scattered across shared folders, the real problem isn’t effort.
It’s workflow architecture.
Let’s break down the packaging labeling mistakes that quietly trigger recalls, regulatory violations, supply chain rejections, and costly relabeling.
1. Mislabeling Allergens and FDA Labeling Violations
Allergen mislabeling continues to be one of the leading causes of product recalls in the United States.
Under the Food Allergen Labeling and Consumer Protection Act (FALCPA), major allergens must be clearly declared. Missing a “Contains” statement, using incorrect terminology, or failing to update labeling after a formulation change constitutes misbranding under FDA regulations and immediately exposes brands to recall risk.
The issue isn’t a lack of awareness.
It’s that version control failures happen quietly.
And compliance gaps stay invisible until they’re public.
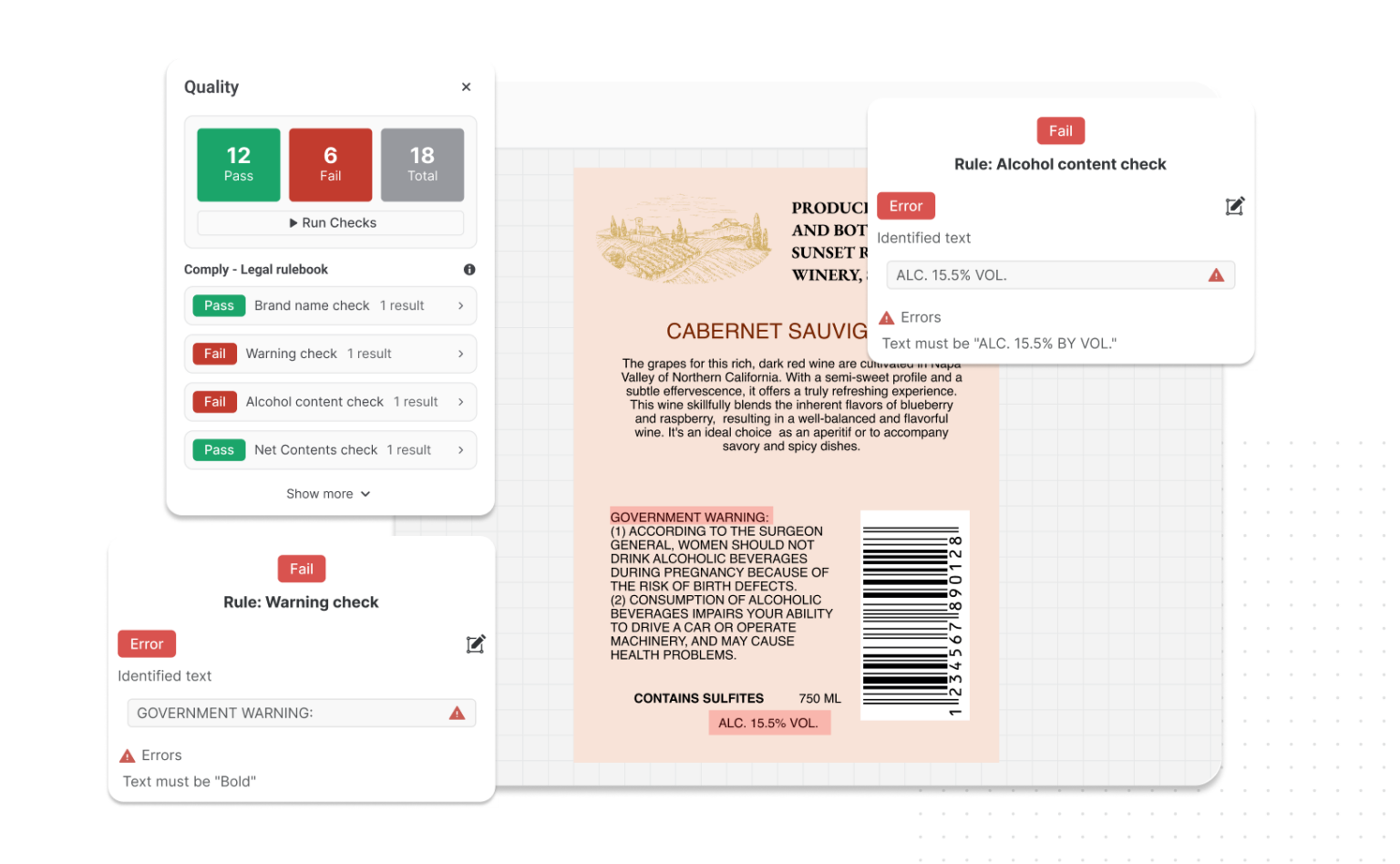
2. Incorrect Net Quantity Statements and Measurement Errors
The net quantity statement isn’t just a design element but regulated declaration governed by the FDA, with strict requirements around unit formatting (oz vs. fl oz), placement on the principal display panel, minimum type size, and approved U.S. measurement standards.
Get any of these wrong, and the product can be deemed misbranded under FDA regulations, even if what’s inside the package is perfectly safe.
3. Nutrition Facts Panel Formatting Errors
A Nutrition Facts panel formatting error occurs when mandatory elements such as serving size, nutrient order, font size, or layout fail to comply with FDA labeling requirements. Even minor formatting deviations can result in misbranding and regulatory enforcement.
Common Nutrition Facts panel formatting errors include:
- Incorrect serving size calculation
- Missing added sugars declaration
- Improper nutrient sequencing
- Failure to meet minimum font size under FDA labeling standards
- Using outdated panel formats
4. Non-Compliant Warning Labels
Warning labels serve a legal purpose: communicating risk clearly and accurately.
When they’re missing, incorrectly formatted, or outdated, they become non-compliant, and that opens the door to enforcement action.
For example, if you plan to sell chemical products in California, you must adhere to Prop 65 labeling requirements.
Under this regulation, products exposing consumers to listed chemicals must carry specific warning language, formatting, and symbols. Incorrect wording or omission of the required symbol can trigger civil penalties and even private enforcement lawsuits.
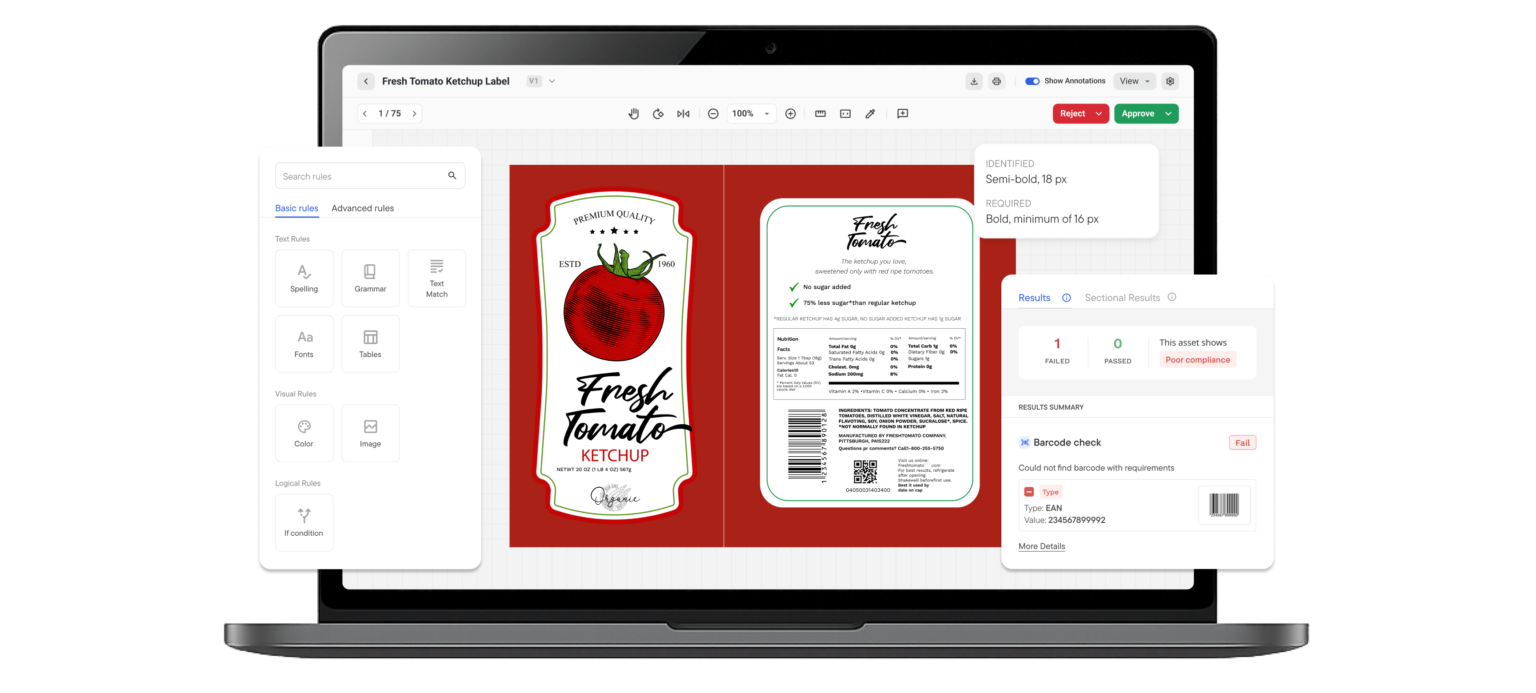
5. Incorrect Barcode and GS1 Formatting Failures
Retailers don’t negotiate barcode or QR code compliance. They reject non-compliant shipments immediately.
GS1 barcode compliance requirements govern:
- Barcode dimensions
- Quiet zone spacing
- Color contrast
- GTIN structure
- Print quality verification
Deviations from GS1 barcode formatting guidelines can prevent products from scanning at distribution centers.
If it doesn’t scan, it doesn’t ship.
6. Multilingual Labeling Compliance Failures
Scaling your business into global markets multiplies risk.
Multilingual labeling compliance requirements vary by region — from bilingual mandates in Canada to country-specific ingredient naming conventions in the EU.
Common issues include:
- Inconsistent translation of regulated terms
- Missing mandatory local disclosures
- Formatting errors across language variants
- Outdated regulatory references
As SKU portfolios grow, manual oversight becomes increasingly fragile. Without centralized governance, version drift becomes inevitable.
7. Expiration Date and Shelf-Life Labeling Compliance Failures
Expiration date labeling compliance impacts both consumer trust and inspection readiness.
If your product expiration or best-by date is:
- Hard to read
- Printed with poor contrast
- In the wrong format
- Misaligned with the actual shelf-life claims
That product can get flagged and pulled long before a consumer notices.
This one is a special blend of legal and operational risk.

Why Packaging Compliance Mistakes Keep Happening
Let’s be honest.
Most organizations blame human error.
But the real issue is workflow architecture.
Most packaging compliance mistakes don’t happen because someone ignored the rules. They happen because the system makes it easy to miss them.
Here’s where things typically break.
1. Manual Approval Processes
If your labeling review process is scattered across spreadsheets, shared folders, and long email threads, risk exposure increases.
Multiple artwork files circulate at once, critical regulatory comments live in inboxes, and “Final_v7_revised_FINAL2.ai” somehow makes it to print.
And when you think things couldn’t get worse, an outdated allergen statement sneaks back in because someone reused a template.
The problem isn’t a lack of review.
Manual approval processes could create version chaos.
When there is no enforced label version control system, compliance breaks quietly.
2. Disconnected Regulatory and Design Workflows
Ask yourself:
- Which label version is currently approved?
- Which SKUs require updated warnings for a new regulation?
- Which markets require multilingual adjustments?
- Can you produce a clean audit trail in minutes?
If those answers require digging through folders, Slack threads, and archived emails, you don’t have centralized compliance visibility.
And without centralized visibility, you cannot systematically reduce labeling errors.
As organizations scale across SKUs, markets, and languages, manual tracking collapses under its own complexity.
3. Lack of Centralized Compliance Visibility
Ask yourself:
- Which label version is currently approved?
- Which SKUs require updated warnings for a new regulation?
- Which markets require multilingual adjustments?
- Can you produce a clean audit trail in minutes?
If those answers require digging through folders, Slack threads, and archived emails, you don’t have centralized compliance visibility.
And without centralized visibility, you cannot systematically reduce labeling errors.
As organizations scale across SKUs, markets, and languages, manual tracking collapses under its own complexity.
Automating Compliance to Prevent Packaging Label Mistakes
By now, one thing should be clear:
You cannot “double-check” your way out of systemic risk.
If packaging compliance mistakes are caused by workflow gaps, then prevention requires workflow control.
This is where labeling compliance automation changes the equation.
Automation embeds regulatory guardrails directly into the packaging lifecycle. Instead of relying on memory, inbox threads, or manual spreadsheets, automated systems enforce structure:
- Pre-print validation ensures required elements are present before files move forward.
- Automated formatting checks flag deviations in Nutrition Facts panels, net quantity placement, and font size requirements.
- Built-in barcode validation verifies GS1 compliance before packaging ever reaches distribution.
- Version control enforcement prevents outdated artwork from re-entering circulation.
- Centralized approvals create a clear chain of accountability.
When automation is handled correctly, it doesn’t slow your team down. It accelerates safe approvals.
And AI-powered packaging label compliance software plays an important role here.
Protect Your Brand with Packaging Label Compliance Software
This is where Comply steps in.
Comply isn’t a basic file repository. It’s purpose-built packaging label compliance software designed to eliminate the structural weaknesses that cause recalls and rejections.
Here’s how Comply strengthens your packaging compliance workflows:
1. Centralized Regulatory Artwork Workflow System
Comply acts as a structured regulatory artwork workflow system, connecting regulatory, marketing, design, and quality teams in one controlled environment.
Instead of approvals happening across email threads and disconnected platforms:
- Every artwork file stays in a governed workspace.
- Comments, edits, and approvals are captured in context.
- Only validated versions can move forward.

2. Enforced Label Version Control System
One of the biggest causes of packaging labeling mistakes is version chaos.
Comply offers a structured label version control system where:
- Approved labels cannot be overwritten.
- Seasonal packaging cannot accidentally replace core SKUs.
- Historical changes are fully traceable.
This prevents outdated ingredient lists, allergen disclosures, or promotional claims from resurfacing months later.
3. Automated Formatting and Regulatory Validation
Compliance drift often happens in small formatting tweaks such as resizing panels, modifying imagery, adjusting typography, or repositioning regulated elements.
With its playbook, Comply integrates automated validation checks to catch:
- Nutrition Facts panel formatting deviations
- Net quantity statement placement issues
- Minimum font size violations
- Missing required fields such as allergen disclosure or warning statements
Instead of discovering issues during a regulatory inspection, teams identify them early in the review process with the help of AI.
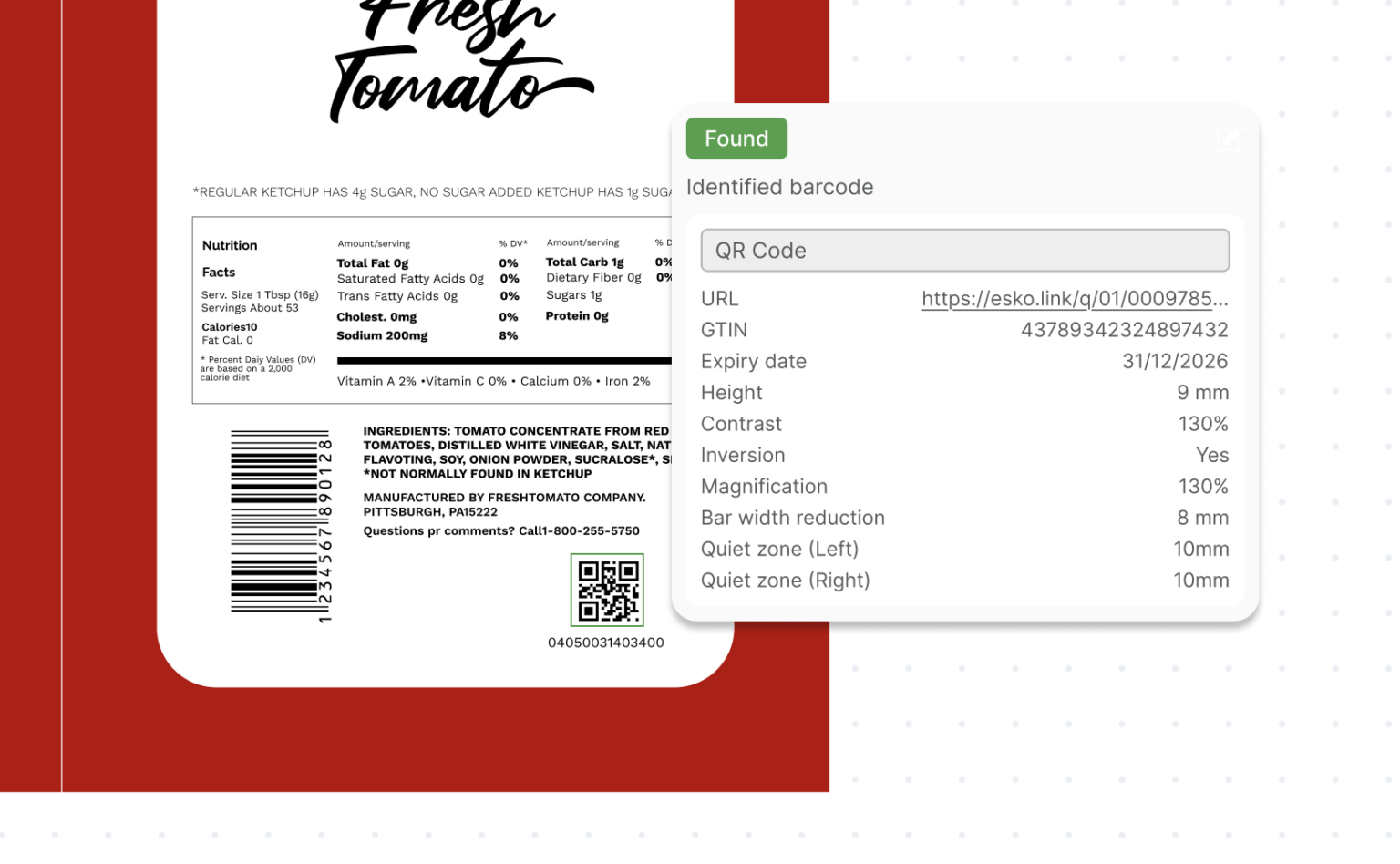
4. Barcode Validation
A beautifully compliant label still fails if the barcode doesn’t scan.
Comply integrates GS1 validation protocols to verify:
- Correct GTIN structure
- Proper barcode dimensions
- Adequate quiet zones
- Contrast and legibility
5. Multi-Market Control
Global expansion and SKU variation introduce layered complexity.
Comply enables centralized control over:
- Region-specific labeling requirements
- Multilingual content alignment
- Market-specific warnings (including Prop 65 and TTB regulations)
- Limited-edition and campaign-specific packaging variant
Instead of managing regional versions manually, teams maintain structured variations within a single governed system that prevents duplication.
6. Audit Trails and Inspection Readiness
When regulators or retail partners request documentation, speed and clarity matter.
Comply maintains comprehensive audit trails detailing:
- Who made changes
- What was modified
- When approvals occurred
- Which version went to print
Final Thoughts
Every major setback usually traces back to something minor that went unnoticed.
A detail overlooked, a change not reflected, or a review that happened too late.
Small cracks in the system create expensive consequences.
If you’re ready to eliminate those cracks with automated label compliance, book a demo of Comply today!
FAQs
What are the most common packaging label compliance mistakes?
The most common packaging compliance mistakes include allergen mislabeling, incorrect net quantity statements, Nutrition Facts formatting errors, non-compliant warning labels, barcode failures, and expiration date labeling issues. These errors often qualify as misbranding under FDA regulations.
What happens if a Nutrition Facts panel is formatted incorrectly?
If a Nutrition Facts panel fails to meet FDA formatting standards, including nutrient order, serving size, or minimum font size, the product may be tagged as misbranded. This can result in warning letters, enforcement action, or mandatory relabeling.
What qualifies as misbranding under FDA labeling laws?
Misbranding occurs when a label is false, misleading, or fails to include required information under the Federal Food, Drug, and Cosmetic Act. This includes inaccurate allergen declarations, improper net quantity statements, and formatting violations in regulated panels.
What are GS1 barcode compliance requirements?
GS1 barcode compliance requirements govern GTIN structure, barcode size, quiet zones, contrast ratios, and print quality standards. Failure to meet GS1 formatting guidelines can lead to retailer rejection and supply chain disruption.
What are Prop 65 labeling requirements?
California Prop 65 labeling requirements mandate specific warning language and symbols when products expose consumers to listed chemicals. Non-compliance may result in civil penalties and private enforcement lawsuits.
How can packaging compliance automation reduce labeling errors?
Packaging compliance automation reduces labeling errors by implementing centralized approvals, automated formatting validation, barcode verification, and version control. By minimizing manual processes, organizations reduce recall risk and generate audit-ready reports.