
Written by Gouri Sasidharan
Content Specialist, Esko
The vitamin gummies market is booming, and it’s not slowing down anytime soon.
In fact, it’s projected to grow from USD 4,593.6 million in 2025 to USD 8,462.2 million by 2035, at a steady CAGR of 6.3%. And honestly, that checks out.
But here’s the thing: those tiny, chewy bears are regulated dietary supplements, and their labels carry some serious weight.
If you’re a brand launching new gummy vitamins to market, their labeling isn’t just packaging. It’s a combination of compliance, transparency, and risk management. And yes, legally binding.
Let’s break down what actually needs to be on a gummy vitamin label.
The Regulatory Framework: Who’s Setting the Rules?
In the U.S., gummy vitamins fall under dietary supplement regulations, governed primarily by:
- The FDA (Food and Drug Administration)
- The Dietary Supplement Health and Education Act (DSHEA)
- The FTC (for marketing and advertising claims)
Unlike conventional foods, supplements must follow specific rules for how nutrients are declared, claims are worded, and information is presented.
Translation?
You can’t just slap “Immune Superpower Gummies” on a bottle and call it a day.
Every claim must be substantiated, and every ingredient must be disclosed. And if you’re making structure/function claims (like “supports bone health”), you must include the FDA disclaimer stating the product is not intended to diagnose, treat, cure, or prevent disease.
Regulations aren’t there to make life harder but to protect consumers and ensure brands compete fairly.
Now let’s get into the real checklist.

The Core Labeling Requirements for Gummies (Non-Negotiables)
If you’re formulating gummy vitamins, this isn’t “nice to have” information. These are mandatory elements. Missing or misplacing even one can trigger regulatory issues, relabeling costs, or, in the worst case, recalls.
Let’s break this down.

1. Statement of Identity
This is the “common name” of the product. It must accurately describe the supplement’s nature, so the consumer isn’t misled. That is, for a product that boosts muscle energy or bone health, you definitely cannot replace it with something trendier like “wellness chews.”
Regulators want consumers to immediately understand the category of product they’re buying.
For gummy vitamins, this usually looks like:
- “Vitamin C Gummies”
- “Multivitamin Gummies”
- “Biotin Gummies”

2. Net Quantity of Contents
Your customers need to understand the value they receive when considering your product.
The Net Quantity of Contents refers to the amount of supplement your package or container has, excluding the weight of the bottle or packaging. It must be expressed accurately in the metric system and the US Customary system. This must be displayed on the Principal Display Panel (PDP) of your label and should include:
- Total number of gummies (e.g., 60 gummies)
- Or weight (e.g., Net Wt. 180 g)
- Or often both
It must be accurate and clearly visible.
If your bottle contains 40 gummies, but the serving size is two gummies per day, that means a 20-day supply. That information must be internally consistent across the entire label.
3. The Supplement Facts Panel
This is the most regulated section of your label. It must follow a standardized FDA format and include:
a) Serving Size
The specific amount of the product intended to be consumed at a time to get the listed nutrient amounts.
b) Servings Per Container
It is automatically calculated based on the total count and serving size.
If you have 60 gummies and a serving size of 2, it must say 30 servings per container.
No rounding games.
c) Active Dietary Ingredients
A list of the vitamins, minerals, or botanicals inside, along with how much of the recommended daily intake they provide. That is,
- The name of the nutrient (e.g., Vitamin D)
- The specific form (e.g., Cholecalciferol)
- The amount per serving (50mcg (2000 IU))
- The % Daily Value (if established)*
*If there is no established Daily Value for a nutrient, it must state:
“Daily Value not established.”
d) % Daily Value (%DV)
The %DV helps consumers understand nutrient contributions relative to daily intake recommendations.
And this must align with current FDA reference values.
If you’re using outdated %DV numbers? That’s a compliance risk.
4. Other Ingredients
The Ingredients List is a mandatory component of your gummy label and is typically placed just below or next to the Supplement Facts panel. While the Supplement Facts highlights only the active nutrients, the Ingredients List covers everything that goes into the product, from actives to supporting ingredients.
All ingredients must be listed in descending order by weight, meaning the ingredient present in the highest quantity appears first.
This list should include:
- Active ingredients (such as vitamins, minerals, or botanical extracts)
- Excipients (including gelling agents, sweeteners, flavorings, acids, oils, and color additives)
In short, if it goes into the gummy, it needs to show up here. No exceptions.
5. Allergens and Warning Statements
This is the “safety net” section. It protects your customers and shields your brand from legal notices.
a) Allergen statement
If your product contains major allergens (milk, soy, wheat, etc.), they must be clearly declared in accordance with FDA labeling regulations.
Gummies can introduce less obvious allergen risks. For example, gelatin may be sourced from fish or pork (relevant for Halal/Kosher claims), while flavorings or coatings can contain hidden allergens like dairy or soy. Even cross-contact risks may need precautionary statements, depending on manufacturing conditions.
This is how you must include your allergen statement:
- “Contains: Soy”
- “Processed in a facility that also handles Milk.
- “May contain traces of Milk and Peanuts”
Allergen statements should be placed immediately after the Ingredients List and clearly emphasized for visibility.
b) Warning Statement
Required cautionary text to prevent misuse, especially since gummies look like candy.
Depending on its formulation, you may need warnings such as:
- “KEEP OUT OF REACH OF CHILDREN”
- “Consult a physician if pregnant or nursing.”
- “Do not exceed recommended dosage.”
6. Structure/Function Claims
If your gummy label says:
- “Supports immune health”
- “Promotes hair growth”
- “Helps maintain bone strength”
Those are structure or function claims.
They are permitted, but they must be truthful, substantiated, and accompanied by the FDA disclaimer:
“These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.”
And remember that you cannot claim to cure, treat, or prevent diseases unless the product is approved as a drug.
There’s a clear regulatory line here.
7. Directions for Use
Before someone starts snacking, they need to know the ground rules. This section ensures that the consumer uses the product safely and effectively.
Your label must include:
- Recommended dosage
- Frequency of consumption
- Age limitations (if applicable)
For example:
“Suggested Use: Take two (2) gummies daily. Chew thoroughly before swallowing.”
8. Manufacturer or Distributor Information
The label must include:
- Company name
- Business address
- Domestic phone number or contact method (if required)
This allows consumers and regulators to contact the responsible party and improves traceability.
If you’re using a contract manufacturer, you still need a clearly identified responsible firm.
9. Lot Number & Expiration Date
Traceability and quality control of your gummies are as important as complying with the FDA regulations.
a) Lot Number
A unique identification code assigned to a specific batch of production.
Example: “Lot #2026-03-A”. If there’s ever a recall, this code is the “Social Security Number” that tracks every bottle from that batch.
b) Expiration (or Best By) Date
The date until which the manufacturer guarantees the product will maintain its full potency and safety.
Gummy vitamins degrade faster than tablets, so this date is non-negotiable for freshness.
Special Labeling Considerations for Children’s Gummies
Gummy supplements are designed to taste good, which is great for compliance, but not always great for portion control (especially with kids).
And if you’ve ever left a bottle of gummy vitamins on the kitchen counter, you already know the problem. Kids don’t see “dietary supplement”. They see colorful fruit snacks.
Because of this, industry groups like the Council for Responsible Nutrition (CRN) have issued additional voluntary labeling guidelines specifically for gummy supplements marketed to children.
These recommendations aim to reduce accidental overconsumption and improve safety.
Here are some of the key labeling considerations.

1. Clearly Indicate the Intended Age Group
Labels should specify the intended users of the product, such as:
- Children
- Adults
- Specific age ranges (e.g., ages 4+)
This helps parents quickly determine whether a product is appropriate for their child.
Without clear age labeling, it becomes easier for products to be used incorrectly.
2. Include Child Safety Warnings
CRN recommends including clear warning statements on gummy supplement labels such as:
- “Keep out of reach of children.”
- “Take only as directed.”
- “Do not exceed recommended intake.”
These statements help reinforce safe use and discourage unsupervised consumption.
3. Choking Hazard Warning for Younger Children
For products intended for children under four years old, CRN recommends including a precautionary statement such as:
This guidance recognizes that gummies can pose a choking risk if swallowed whole, a reason why labels should instruct children to fully chew each gummy before swallowing.
Consider Child-Deterrent Packaging
While not strictly a labeling rule, CRN also encourages companies to consider child-deterrent packaging for gummy supplements.
Since gummies resemble candy, packaging that prevents unsupervised access adds another layer of protection for families. That’s why labels for children’s supplementary gummies need extra clarity.
Design and Legibility Requirements for Your Vitamin Gummies
The FDA doesn’t just care about what you say; they care about how it looks. If your label is so cluttered that the important stuff is hidden, you’re asking for trouble.
a. Font style
Your customer didn’t sign up for squinting eyes at your labels. It is best advised to use Arial or Helvetica on your vitamin gummy labels, which improves readability.
b. Type Size (The 1/16th Rule):
Most mandatory information must be printed in a type size of at least 1/16th of an inch based on the height of a lowercase “o.”
c. Contrast and Clear Backgrounds:
Any information you include must be conspicuous. This means you need a high contrast between the text and the background. For example, adding white text on a light-yellow background is a classic “no-no”. Black text on a White background would be ideal.
d. The “Supplement Facts” Box Formatting:
The FDA has a very specific “look” for this box. It needs those heavy black borders and specific bolding to separate sections. You’ll notice that the words “Supplement Facts” are always the largest and boldest part of that panel.
How Esko Comply Can Help Nail Your Packaging Labels
Navigating the maze of FDA regulations is enough to give anyone a headache. That’s where the AI-led artwork compliance software, Comply, plays a critical role in simplifying your packaging compliance.
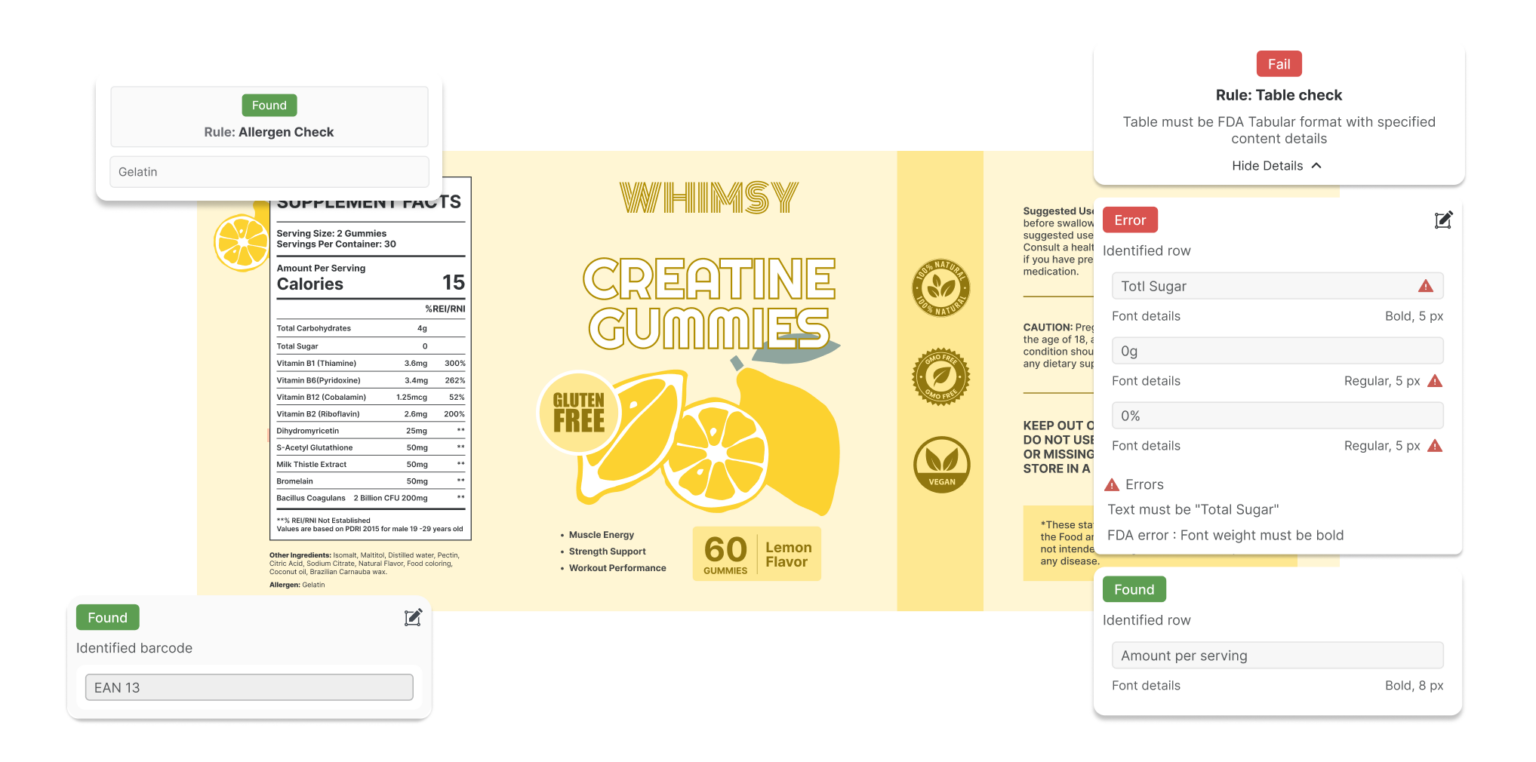
Automated Compliance Checks
Comply helps ensure your labels align with current regulatory requirements by automatically validating key elements like ingredient declarations, allergen statements, and mandatory disclosures. It reduces the risk of manual error and keeps you audit-ready.
Centralized Ingredient & Claim Management
Manage all your ingredient data, claims, and regulatory content in one place. This makes it easier to maintain consistency across products, avoid duplication, and quickly update labels when formulations or regulations change.
Multi-Market Labeling Support
If you’re looking to expand your business globally or find that managing artwork lifecycle is becoming complex, Comply can help you meet labeling requirements for various regions, such as the U.S., EU, or UK, with its flexible playbook. This ensures that your products remain compliant wherever they are sold, without the need to start from scratch.
Reduced Time-to-Market
By automating repetitive tasks and minimizing back-and-forth between teams, Comply helps you move from formulation to shelf faster without compromising accuracy.
Improved Collaboration & Version Control
Keep regulatory, NPD, design, and packaging teams aligned with shared workflows and version tracking. No more confusion over which label version is the “final” one.
Transparency is the Best Policy
Ultimately, your customer needs to make informed decisions by clearly understanding the labeling requirements.
For your brand, mastering those requirements means protecting your reputation, avoiding regulatory trouble, and building trust.
If you’re looking to simplify your label compliance process and get to market faster, check out Comply!







