Written by Mitha Shameer
Content Specialist, Esko
Here’s a number worth sitting with: in 2025, the FDA issued 251 food and beverage recall events. That’s roughly five recalls every single week. Products pulled from shelves. Supply chains thrown into chaos. Brand managers fielding calls they never want to get.
Now here’s the number that should really give you pause.
For brands that invest heavily in food safety programs, supplier audits, and quality systems, that statistic should sting a little. Because it means the most common source of recalls in 2025 wasn’t a manufacturing failure. It was a documentation and labeling failure — the kind that happens after the product has been made correctly, and before it gets to the shelf.
That’s a very specific, very solvable problem. Let’s talk about it.
The 2025 Recall Landscape, In Full
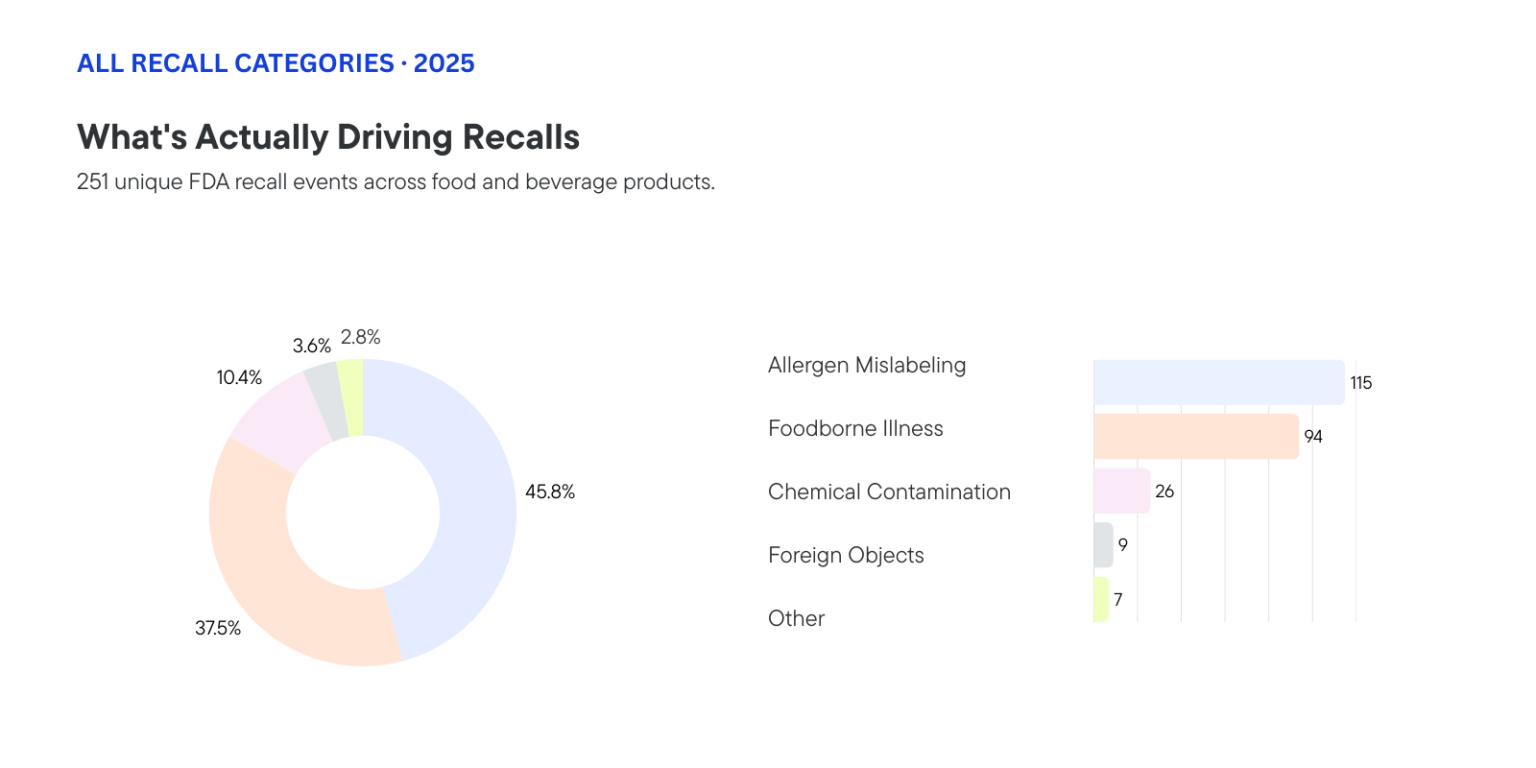
Before we get to solutions, it’s worth looking at the full picture. Because while allergens dominate the headline number, the data tells a few different stories at once.
- Allergen mislabeling: 115 recalls (45.8%)
- Foodborne illness: 94 recalls (37.5%) — Listeria, Salmonella, Clostridium botulinum, E. coli
- Chemical/radiological contamination: 26 recalls (10.4%) — including a striking cluster of Cesium-137 recalls in frozen shrimp
- Foreign objects: 9 recalls (3.6%) — plastic, stones, glass, metal, wood
- Other: 7 recalls (2.8%) — choking hazards, mislabeled alcohol content
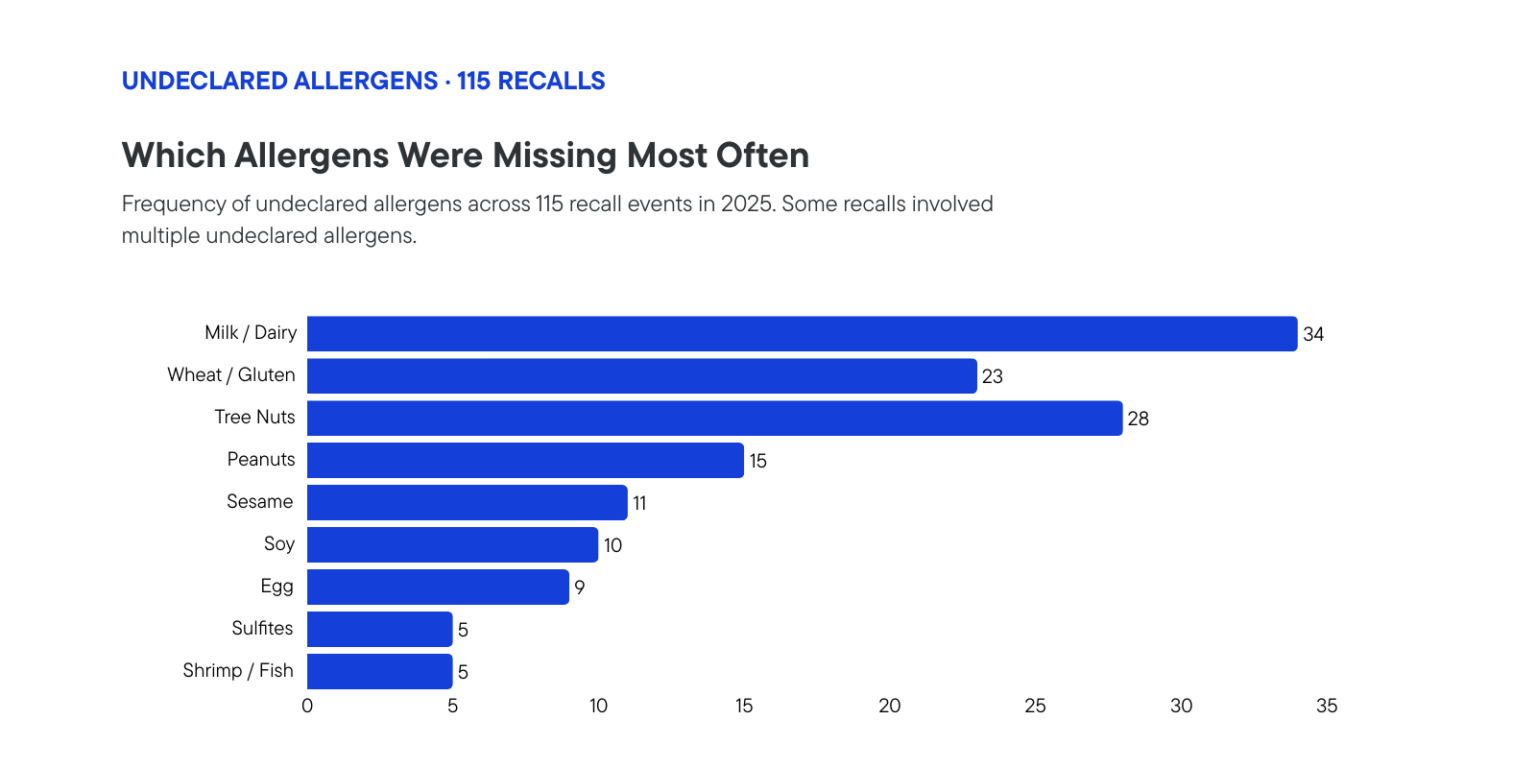
While foodborne illness outbreaks often dominate headlines, it is allergen mislabeling that dominates the recall register. Milk and dairy were the most frequently undeclared allergens, appearing in around 30% of allergen-related recalls, followed by tree nuts, wheat, peanuts, sesame, and soy. The recalls span every product category, from ice cream and chocolate bars to tortilla chips, pasta, and infant formula.
Listeria monocytogenes was the most common foodborne pathogen, driving 42 recalls, turning up in cheese, pasta salads, mushrooms, and fresh produce. Salmonella followed with 35, spiking hard in May and June during a widespread cucumber contamination event that swept through multiple retail brands simultaneously.
And then there’s Cesium-137, a radioactive contamination cluster that triggered 11 recalls of frozen shrimp products across multiple brands and retailers. That one was impossible to prevent at the label stage, and a useful reminder that not every recall has a clean solution.
This Wasn't a Small-Brand Problem
One of the more humbling things about the 2025 allergen recall data is who it hit. Among the 115 allergen recalls were products from nationally recognized manufacturers: frozen snacks, condiments, ice cream, salad kits, and fast-food staples.
- A cracker sandwich recalled twice for the same undeclared peanut.
- A tortilla chip with undeclared milk.
- A frozen burger slider with undeclared milk and soy.
- A salad kit missing sesame and soy.
- A condiment dressing missing four allergens at once: peanut, soy, sesame, and wheat.
- An ice cream range missing three nut allergens from its declaration.
Where the Gap Actually Lives
Here’s how most allergen mislabeling incidents actually unfold. It’s rarely as dramatic as it sounds.
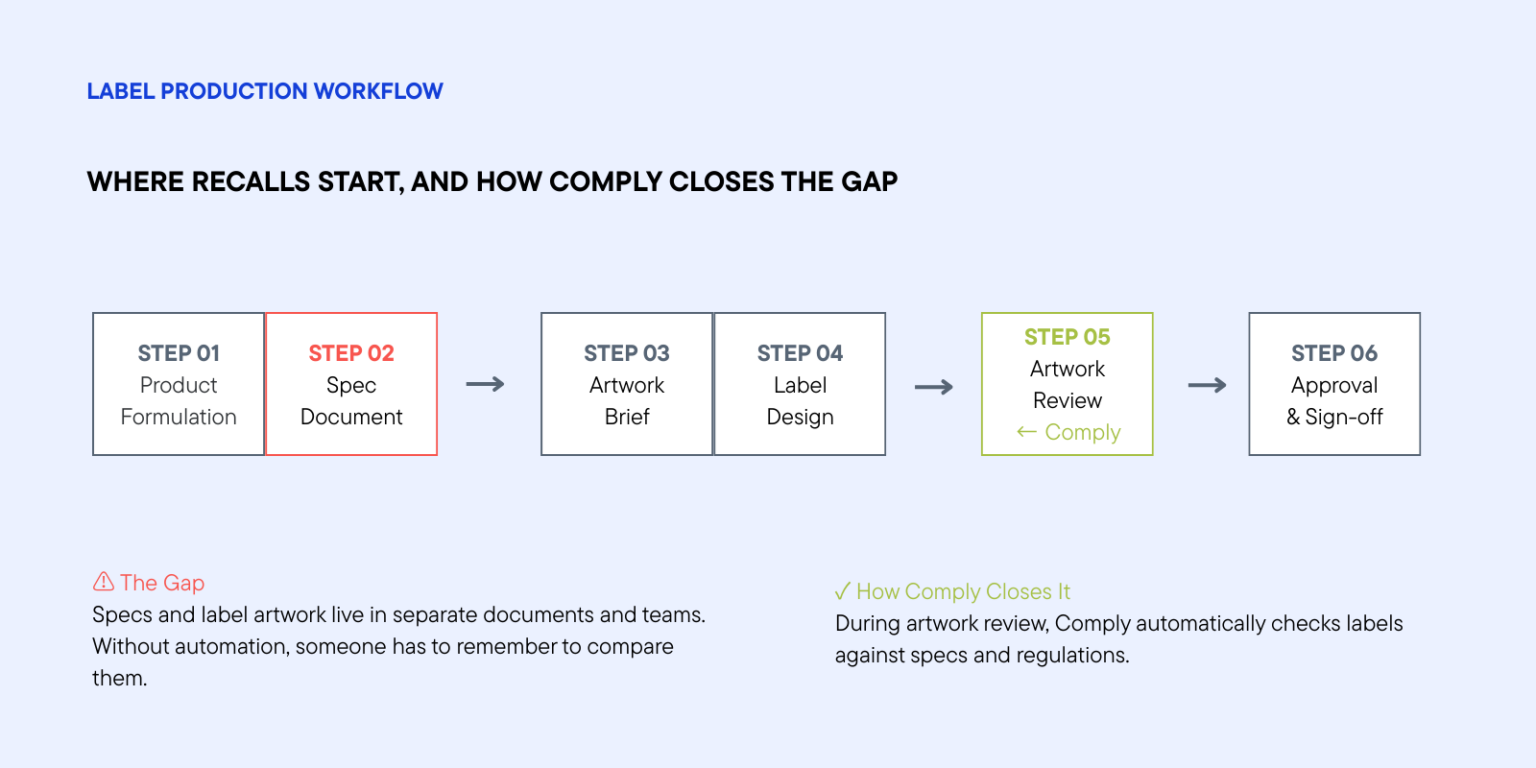
A product formulation changes with a new ingredient, a supplier swap, or a recipe tweak. The update gets recorded in the specification document. But the label artwork was created six months ago, lives in a shared drive somewhere, and is managed by a different team. The connection between the spec and the label exists in theory. In practice, it depends on someone remembering to check.
Or: a product is being reformulated for a new market. The core recipe stays the same, but the regulatory requirements for allergen declaration are different. Someone familiar with one market’s rules approves the label. No one flags that the rules are different here.
Or: the label is just wrong. A copy-paste error, an outdated version, a last-minute change that didn’t make it through the approval chain.
In each scenario, the failure point is the same: there’s no systematic, automated check that compares what the label says against what it’s supposed to say. Human reviewers are doing their best, but they’re working against deadline pressure, version confusion, and the sheer volume of SKUs that modern food brands manage.
THE STRUCTURAL PROBLEM
Most brands have good documentation. Most brands have approval workflows. What most brands don’t have is an intelligent layer that automatically verifies the label against both the regulatory requirements for that market and the product’s own specification, before it goes to print.
What Better Actually Looks Like
An artwork management system is the obvious starting point. It gives you version control, structured approval workflows, and a single source of truth for label artwork — all the infrastructure you need to manage label production at scale without things falling through the cracks. But an AMS alone doesn’t close the gap. It manages the process but doesn’t verify the content.
The next step is embedding intelligent compliance checking directly into that review process, so that every time a reviewer opens a file, the system has already done the heavy lifting. Tools like Comply, from Esko, are built specifically for this: AI-assisted checks that run inside your existing packaging workflow, comparing the artwork against market regulations and the product specification simultaneously.
The practical effect is straightforward. A reviewer opens a file and sees a prioritized list of issues already flagged. They can approve, reject, or annotate with context. Comply reads the label and checks it against two things simultaneously:
1. The market specification
Every market has its own rules. What allergens need to be declared, how they need to be phrased, where they need to appear, what font size they need to be. It varies by country, retailer, and product category. Comply holds these specifications and checks your label against them automatically.
For brands selling across multiple markets — and most brands of any size are — this is where things get complicated fast. A label that clears every check for the US market may fail three checks for the EU. Comply flags these discrepancies before a single label goes to print.
2. The product specification document
This is the closer-to-home check, and it’s the one that would have caught a lot of the 2025 recalls.
Comply reads the product spec — the document that defines what’s in the product, what allergens are present, what claims can be made — and compares it against the label artwork. If the spec says the product contains milk and the label doesn’t declare milk, that’s a failure. Comply catches it, and the artwork doesn’t get approved.
It doesn’t matter how many SKUs you’re managing or how many markets you’re selling into. The check happens every time, on every label, without exception.
Comply doesn’t replace your artwork team or your QA process. It gives them a safety net that doesn’t get tired, doesn’t miss things under deadline pressure, and doesn’t rely on someone remembering to check.
Prevention Is Cheaper Than Recall
A product recall is never just a logistics problem. FDA-regulated recalls require formal notification, product retrieval, root cause investigation, and corrective action reporting. Beyond the direct costs, which for large-volume products can run into millions, there is the reputational cost, the retailer relationship cost, and in allergen cases, the very real human cost to consumers whose health depends on accurate labeling.
The 2025 data shows that the brands most at risk are not necessarily the smallest or least resourced. The problem is structural, not a matter of intent.
Embedding intelligent compliance checking into the artwork approval workflow, through tools like Comply, addresses the problem structurally. It makes allergen verification a systematic, automated step in every label review rather than an item on a checklist that depends on individual attention and memory.
Recalls trigger regulatory investigations, erodes retailer trust, damages brand equity, and in the worst cases, puts vulnerable consumers in hospital. The good news is that the majority of the 2025 allergen-related recalls share a common root cause that modern technology is now well-equipped to prevent.
The Bottom Line
The 2025 FDA recall data is a useful reality check. Nearly half of all recalls trace back to a label that was missing something it should have said. That is a solvable problem.
An artwork management system gives you the workflow infrastructure to manage label production properly. Comply adds an intelligent compliance layer that checks every label against market specifications and product specs automatically, before print.
In a year where 115 brands learned that lesson the hard way, the case for getting ahead of it has never been clearer. Explore Comply today.
A quick note on the data: this analysis covers FDA recall records from January through December 2025. We have included pet food recalls but excluded dietary supplements and medical devices. We reviewed the full dataset carefully to remove duplicates, but given the volume involved, a small margin of error should be assumed. So, treat these numbers as directionally accurate rather than definitive.