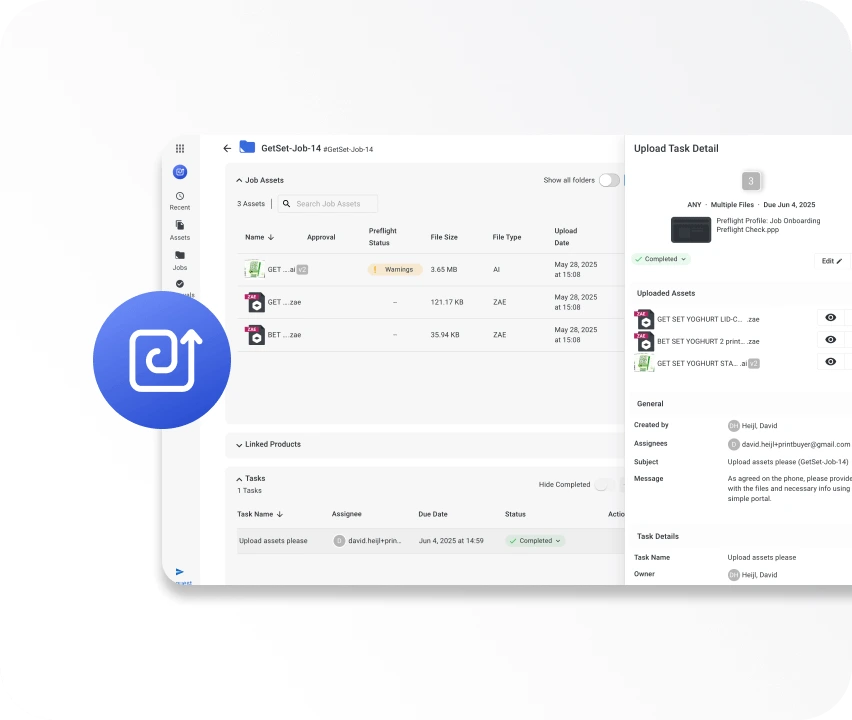
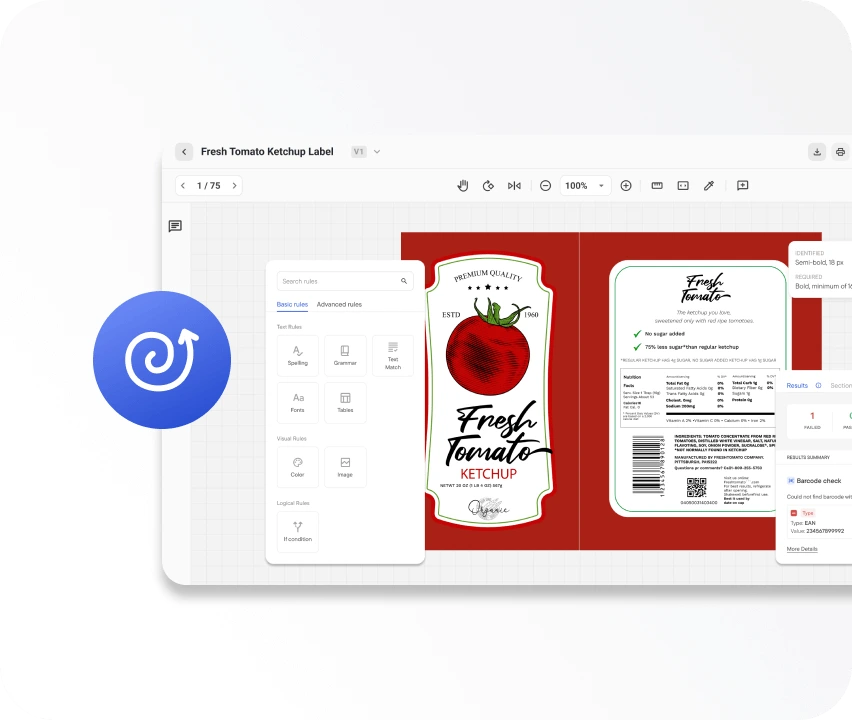
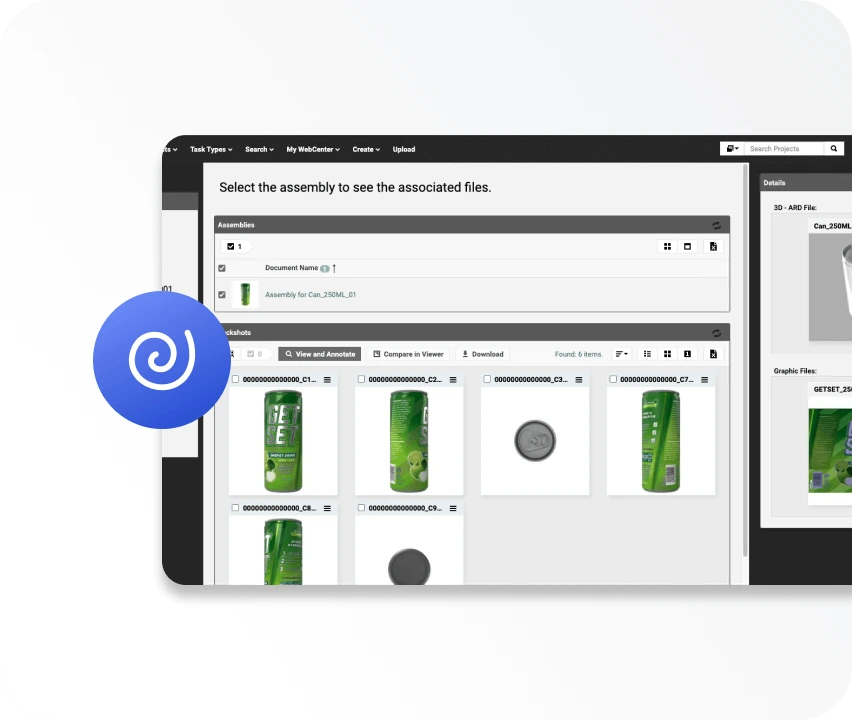
WebCenter
AI-Enabled Artwork and Packaging Management
Whether you’re a packaging converter, print service provider, growing or enterprise brand, there’s a WebCenter solution for you.
Find Your Perfect Solution
| WebCenter Pack | WebCenter Go | WebCenter Enterprise | |
|---|---|---|---|
The Benefits of WebCenter as an Artwork Management System